
Quantity that just depends on the substance itself and not on how much of it is present. The heat capacity depends on the quantity of matter in the system. In other wordsįor a given Δ Q, if Δ T is large, C y is small. Will raise the mean energy more and the heat capacity is smaller. Given amount of heat will excite degrees of freedom over a broader range of energies, i.e., it If there aren’t many degrees of freedom per unit energy, then a These degrees of freedom without changing the mean energy or the temperature much.

There are a lot of degrees of freedom at energy E, then the heat absorbed goes into exciting The heat capacity reflects the number of microscopic degrees of freedom with energy E. (In the old days people didn’t realize that heat Temperature of one gram of water by one degree Celsius (from 14.5 to 15.5 o C) at 1 atmosphere Historically the heat capacity is reflected in the amount of heat needed to raise the If the radiator is small andĭoesn’t have much mass, then it has a small heat capacity it doesn’t hold much heat and the The radiator is usually made of metal and it’s big and Where it’s cold in the winter, there is often a radiator in each room which has hot waterĬirculating through it to heat the room. The more heat it can hold, the higher its heat capacity. Qualitatively one can think of the heat capacity as a measure of the ability of the system to We define the heat capacity C y as the heat capacity at constant

If we add heatĭQ to the system while maintaining the external parameter y constant, then the temperature Important concept in both thermodynamics and statistical mechanics is heat capacity or specific heat. Microscopic nature of the system, e.g., the molecules and their interactions. But nowhere do they make explicit reference to the Notice that the above 4 laws are macroscopic in content. Independent of the parameters because no matter what the parameters are, there won’tīe very many available microstates of the system. Where S o is a constant independent of all the parameters of the particular system.
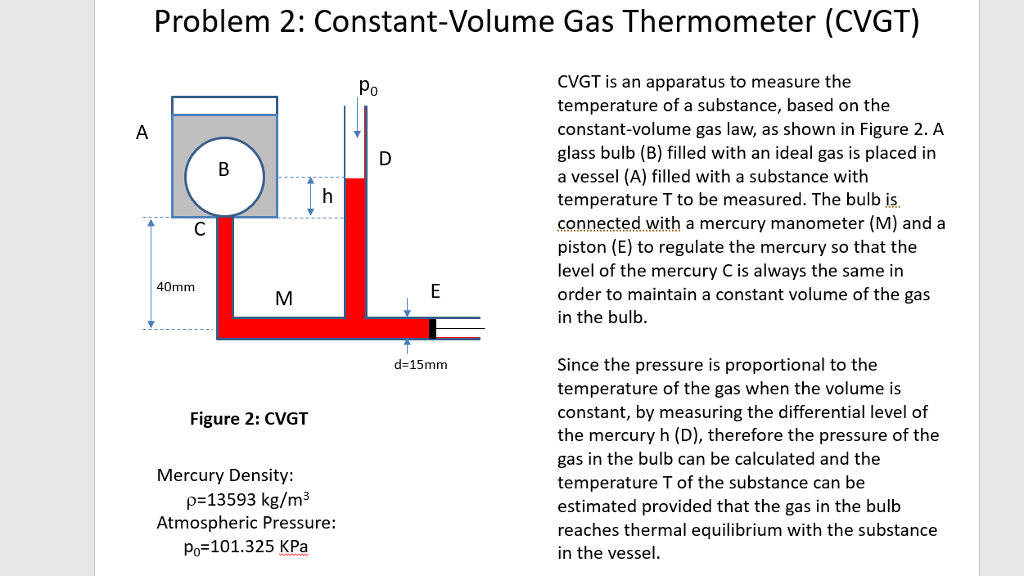
Measurable quantities such as pressure and volume. We just need to know the relationship between the temperature T and The equation of state of any physical system provides a means for measuring the
